|
However, a pair of atoms may need to share more than one pair of electrons in order to achieve the requisite octet. Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: Double and Triple BondsĪs previously mentioned, when a pair of atoms shares one pair of electrons, we call this a single bond. To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. The transition elements and inner transition elements also do not follow the octet rule: Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule. 
These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). 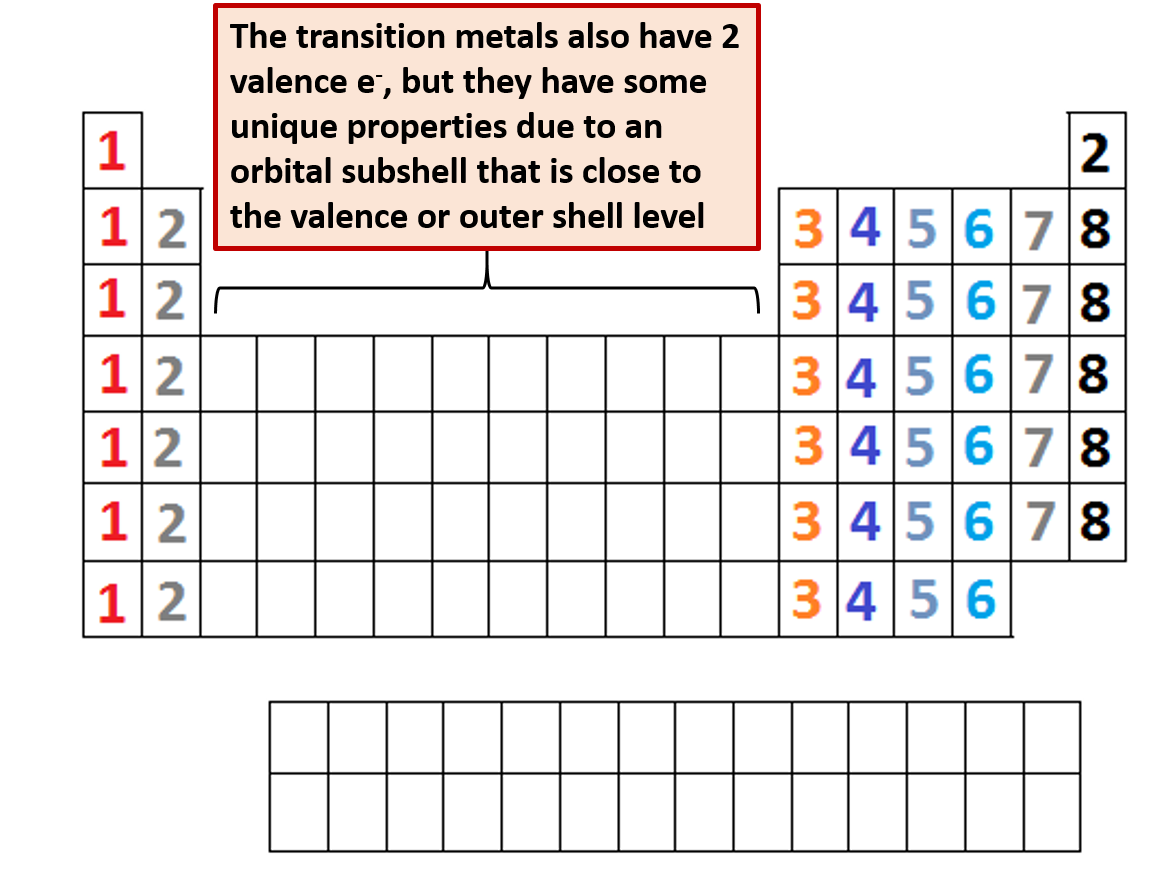
For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). The tendency of main group atoms to form enough bonds to obtain eight valence electrons is known as the octet rule. This allows each halogen atom to have a noble gas electron configuration. The other halogen molecules (F 2, Br 2, I 2, and At 2) form bonds like those in the chlorine molecule: one single bond between atoms and three lone pairs of electrons per atom. Each Cl atom interacts with eight valence electrons: the six in the lone pairs and the two in the single bond. A dash (or line) is sometimes used to indicate a shared pair of electrons:Ī single shared pair of electrons is called a single bond. The Lewis structure indicates that each Cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). For example, when two chlorine atoms form a chlorine molecule, they share one pair of electrons: 
We also use Lewis symbols to indicate the formation of covalent bonds, which are shown in Lewis structures, drawings that describe the bonding in molecules and polyatomic ions. The total number of electrons does not change. Oxygen will gain 2 electrons.Figure 4.11 Cations are formed when atoms lose electrons, represented by fewer Lewis dots, whereas anions are formed by atoms gaining electrons. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. 
Write the electron configuration of oxygen atom (Z=8). In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. On the left, the chlorine atom has 17 electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
